Imaging of the centromedian thalamic nucleus using 3D fast gray matter acquisition T1 inversion recovery sequence in 3T MRI in epilepsy
Keywords:
Centromedian thalamic nucleus, epilepsy, fast gray matter acquisition T1 inversion recovery, magnetic resonance imaging, thalamusAbstract
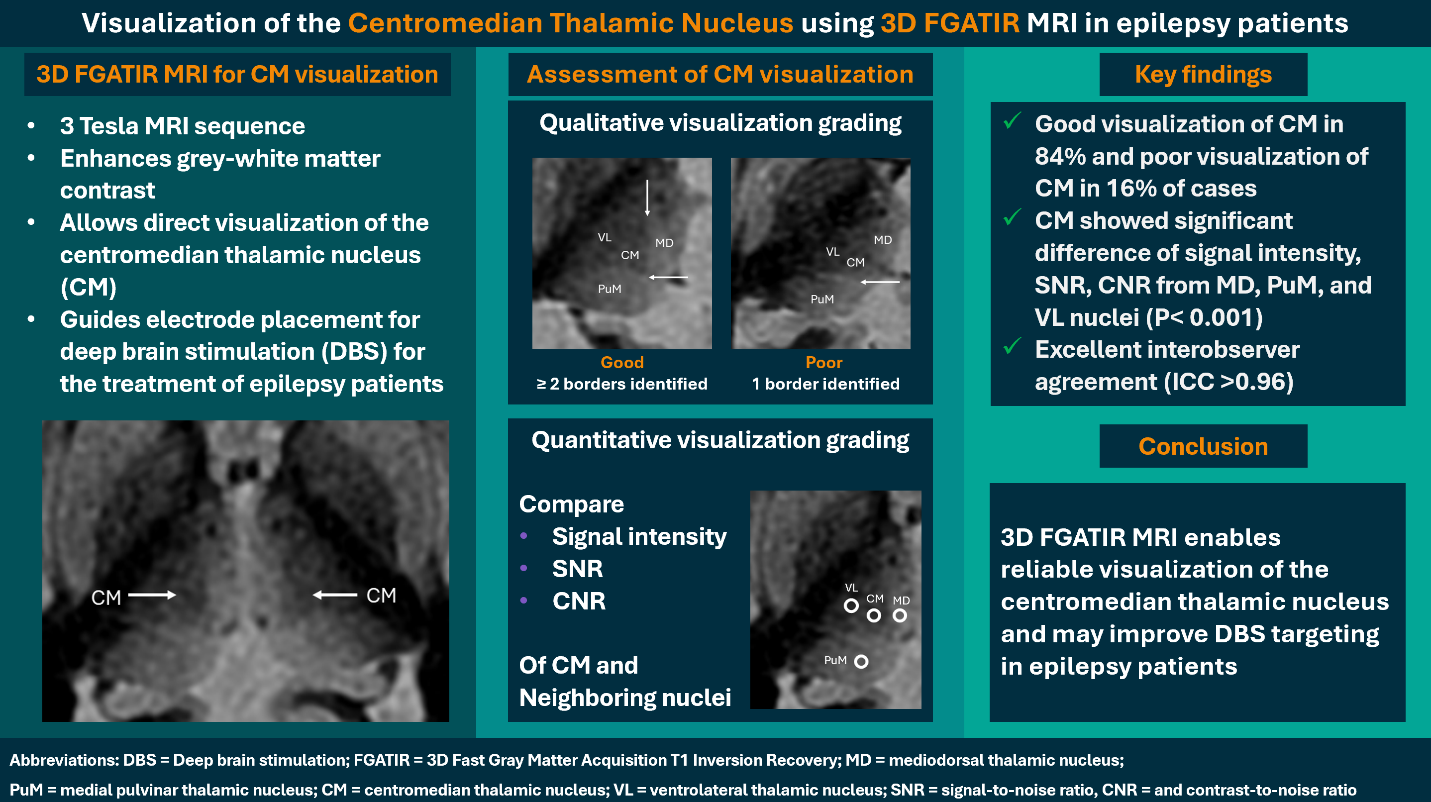
Background: The centromedian thalamic nucleus (CM) is an emerging deep brain stimulation (DBS) target for neurological disorders, including drug-resistant epilepsy. Traditional DBS localization relies on predefined stereotactic coordinates, which may be imprecise. Innovative magnetic resonance imaging (MRI) sequences enhance gray–white matter contrast, potentially allowing direct CM visualization and improving accuracy in DBS planning and targeting.
Objective: This study evaluated the 3D fast gray matter acquisition T1 inversion recovery (FGATIR) MRI sequence for visualizing the CM in patients with epilepsy.
Methods: This prospective IRB-approved descriptive study included adult patients with epilepsy who underwent a 3D FGATIR MRI sequence at 3 Tesla. Two neuroradiologists independently assessed CM visualization using qualitative grading and quantitative measurements of signal-to-noise ratios (SNR) and contrast-to-noise ratios (CNR) of the CM and three neighboring nuclei. These nuclei included the mediodorsal (MD), medial pulvinar (PuM), and ventrolateral (VL) nuclei in each thalamus. Interobserver agreement for quantitative assessment was evaluated with the intraclass correlation coefficient (ICC).
Results:A total of 16 patients with epilepsy (mean age 42.8 ± 21.3 years, 44.0% male) were included. Qualitative assessment revealed good CM visualization in 84.0% and poor CM visualization in 16.0% of cases. Mean signal intensities and SNRs differed significantly between the CM and other neighboring thalamic nuclei (P < 0.001). CNRs of the CM relative to the MD, PuM, and VL varied across the thalami. Interobserver agreement was excellent, with ICC > 0.96 for all measurements.
Conclusion: The 3D FGATIR MRI sequence is a practical tool to visualize the CM. Its adoption in clinical practice could potentially improve DBS planning.
Downloads
References
Warren AE, Dalic LJ, Thevathasan W, Roten A, Bulluss KJ, Archer J. Targeting the centromedian thalamic nucleus for deep brain stimulation. J Neurol Neurosurg Psychiatry 2020;91:339-49.
https://doi.org/10.1136/jnnp-2019-322030
Ilyas A, Pizarro D, Romeo AK, Riley KO, Pati S. The centromedian nucleus: anatomy, physiology, and clinical implications. J Clin Neurosci 2019;63:1-7.
https://doi.org/10.1016/j.jocn.2019.01.050
Singh H, Stamm M, Warren AEL, Kulsomphob A, Jha R, Bhatia N, et al. Optimizing indirect targeting of the centromedian nucleus for deep brain stimulation by incorporating third ventricular anatomy. J Neurosurg 2024;142:299-310.
https://doi.org/10.3171/2024.4.JNS24286
Cukiert A, Lehtimäki K. Deep brain stimulation targeting in refractory epilepsy. Epilepsia 2017;58 Suppl 1:80-4.
https://doi.org/10.1111/epi.13686
Chan DT, Zhu XL, Yeung JH, Mok VC, Wong E, Lau C, et al. Complications of deep brain stimulation: a collective review. Asian J Surg 2009;32:258-63.
https://doi.org/10.1016/S1015-9584(09)60404-8
Li J, Li Y, Gutierrez L, Xu W, Wu Y, Liu C, et al. Imaging the centromedian thalamic nucleus using quantitative susceptibility mapping. Front Hum Neurosci 2020;13:447.
https://doi.org/10.3389/fnhum.2019.00447
Saranathan M, Iglehart C, Monti M, Tourdias T, Rutt B. In vivo high-resolution structural MRI-based atlas of human thalamic nuclei. Sci Data 2021;8:275.
https://doi.org/10.1038/s41597-021-01062-y
Middlebrooks EH, Tao S, Zhou X, Greco E, Westerhold EM, Tipton PW, et al. Synthetic inversion image generation using MP2RAGE T1 mapping for surgical targeting in deep brain stimulation and lesioning. Stereotact Funct Neurosurg 2023;101:326-31.
https://doi.org/10.1159/000533259
Eckert U, Metzger CD, Buchmann JE, Kaufmann J, Osoba A, Li M, et al. Preferential networks of the mediodorsal nucleus and centromedian-parafascicular complex of the thalamus--a DTI tractography study. Hum Brain Mapp 2012;33:2627-37.
https://doi.org/10.1002/hbm.21389
Sudhyadhom A, Haq IU, Foote KD, Okun MS, Bova FJ. A high resolution and high contrast MRI for differentiation of subcortical structures for DBS targeting: the Fast Gray Matter Acquisition T1 Inversion Recovery (FGATIR). Neuroimage 2009;47 Suppl 2:T44-T52.
https://doi.org/10.1016/j.neuroimage.2009.04.018
Joshi CN, Karakas C, Eschbach K, Samanta D, Auguste K, Desai V, et al. Pediatric neuromodulation for drug‐resistant epilepsy: Survey of current practices, techniques, and outcomes across US epilepsy centers. Epilepsia Open 2024;9:785-92.
https://doi.org/10.1002/epi4.12902
Rijks N, Potters WV, Dilai J, De Bie RMA, de Win M, van der Zwaag W, et al. Combining 7T T2 and 3T FGATIR: from physiological to anatomical identification of the subthalamic nucleus borders. J Neurol Neurosurg Psychiatry 2022;93:1019-20.
https://doi.org/10.1136/jnnp-2021-326710
Neudorfer C, Kroneberg D, Al‐Fatly B, Goede L, Kübler D, Faust K, et al. Personalizing deep brain stimulation using advanced imaging sequences. Ann Neurol 2022;91:613-28.
https://doi.org/10.1002/ana.26326
Grewal SS, Middlebrooks EH, Kaufmann TJ, Stead M, Lundstrom BN, Worrell GA, et al. Fast gray matter acquisition T1 inversion recovery MRI to delineate the mammillothalamic tract for preoperative direct targeting of the anterior nucleus of the thalamus for deep brain stimulation in epilepsy. Neurosurg Focus 2018;45:E6.
https://doi.org/10.3171/2018.4.FOCUS18147
Morishita T, Higuchi MA, Kobayashi H, Abe H, Higashi T, Inoue T. A retrospective evaluation of thalamic targeting for tremor deep brain stimulation using high-resolution anatomical imaging with supplementary fiber tractography. J Neurol Sci 2019;398:148-56.
https://doi.org/10.1016/j.jns.2019.01.025
Morel A. Stereotactic atlas of the human thalamus and basal ganglia. Boca Raton: CRC Press; 2007.
https://doi.org/10.3109/9781420016796
National Electrical Manufacturers Association. Determination of Signal-to-Noise Ratio (SNR) in diagnostic magnetic resonance imaging. Rosslyn, VA: National Electrical Manufacturers Association; 2008.
Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 2016;15:155-63.
https://doi.org/10.1016/j.jcm.2016.02.012
Tourdias T, Saranathan M, Levesque IR, Su J, Rutt BK. Visualization of intra-thalamic nuclei with optimized white-matter-nulled MPRAGE at 7 T. Neuroimage 2014;84:534-45.
https://doi.org/10.1016/j.neuroimage.2013.08.069
Su JH, Thomas FT, Kasoff WS, Tourdias T, Choi EY, Rutt BK, et al. Thalamus Optimized Multi Atlas Segmentation (THOMAS): fast, fully automated segmentation of thalamic nuclei from structural MRI. Neuroimage 2019;194:272-82.
https://doi.org/10.1016/j.neuroimage.2019.03.021
Morel A, Magnin M, Jeanmonod D. Multiarchitectonic and stereotactic atlas of the human thalamus. J Comp Neurol 1997;387:588-630.
https://doi.org/10.1002/(SICI)1096-9861(19971103)387:4<588::AID-CNE8>3.0.CO;2-Z
Viera AJ, Garrett JM. Understanding interobserver agreement: the kappa statistic. Fam Med 2005;37:360-3.
Feinstein AR, Cicchetti DV. High agreement but low kappa: I. The problems of two paradoxes. J Clin Epidemiol 1990;43:543-9.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Chulalongkorn Medical Journal

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.









